Reductive Action of Light-Generated Electrons or Hot Spots? Elucidating the Mechanism Behind World-Leading CO2 Photocatalytic Fuel Conversion Activity
A research group from Chiba University and the Chengdu Institute of Biology has clarified the distinct roles of light-generated electrons and hot spots in the photocatalytic conversion of CO₂ to methane. They developed a novel Ni–Ru–ZrO₂ catalyst that achieves world-leading CO₂ photo-methanation rates, demonstrating that Ru addition facilitates CO₂ adsorption, allowing the reaction to proceed via light-induced heating. This breakthrough offers critical guidance for developing highly efficient CO₂ photoreductive catalysts.
📋 Article Processing Timeline
- 📰 Published: April 3, 2026 at 19:00
- 🔍 Collected: April 3, 2026 at 10:31
- 🤖 AI Analyzed: April 17, 2026 at 08:56 (334h 24m after Collected)
A research group comprising Masahito Sasaki (Master's student), Tomoki Oyumi (Doctoral student), and Keisuke Hara (at the time of research) from the Graduate School of Engineering, Chiba University; Professor Yasuo Izumi from the Graduate School of Science, Chiba University; and Associate Professor Hongwei Zhang from the Chengdu Institute of Biology, Chinese Academy of Sciences, has succeeded in clearly identifying and specifying the roles of "light-generated electrons (Note 1)" and "hot spots (Note 2)" in the photocatalytic reaction that converts carbon dioxide (CO₂) into fuels such as methane (CH₄), the main component of natural gas and city gas, which has been a long-standing mystery.
Furthermore, they developed a Ni–Ru–ZrO2 catalyst combining nickel (Ni), ruthenium (Ru), and zirconium dioxide (ZrO2), achieving the world's highest conversion rate of CO₂ to CH₄ at a level of 10 millimoles per gram of catalyst per hour. This achievement, which clarifies the photocatalytic mechanism according to the temperature of the photocatalyst, is expected to provide guidelines for improving the efficiency of CO₂ photoreductive catalysts. This research was published on March 20, 2026 (US time) in the Journal of the American Chemical Society, a publication of the American Chemical Society.
(Paper here: 10.1021/jacs.5c17533)
■ Research Achievements
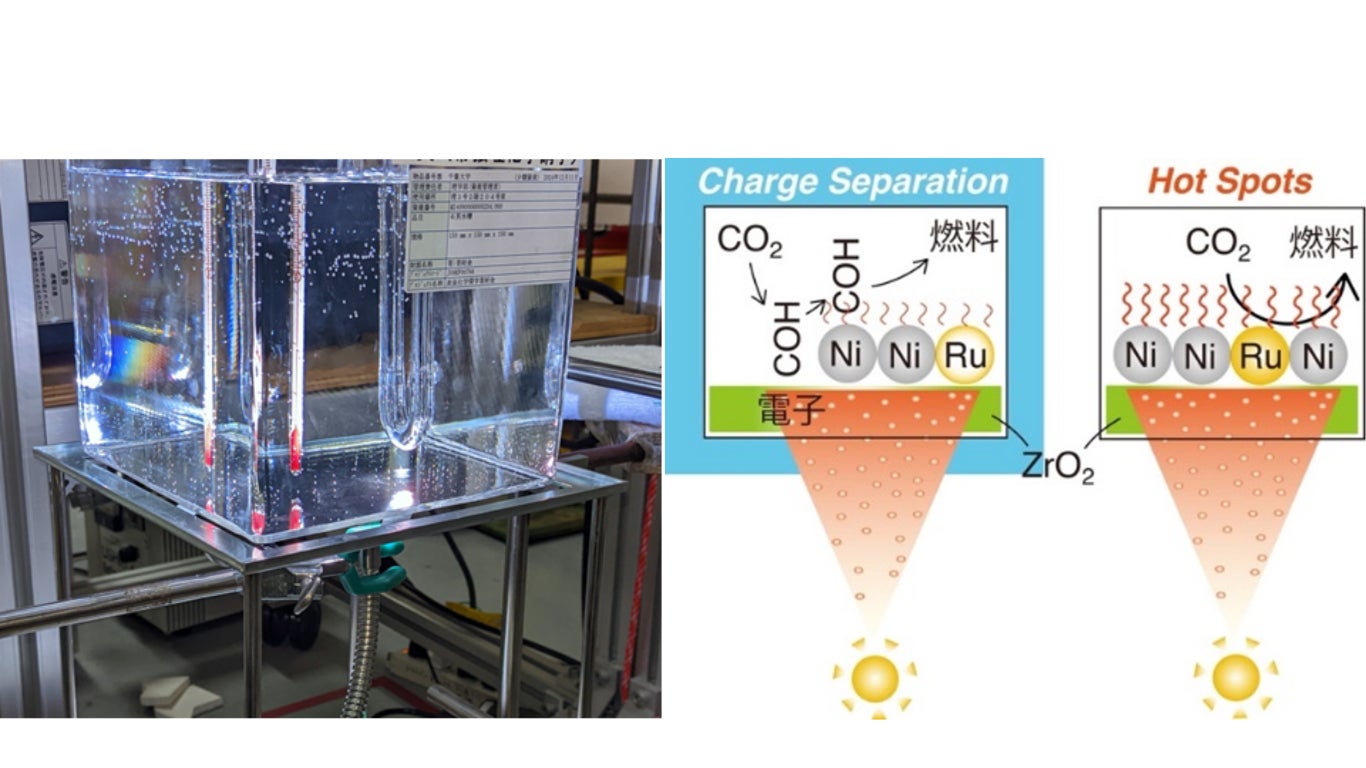
If CO₂ can be converted into fuels and chemical substances through renewable methods using solar energy, a new carbon-neutral cycle can be created. A major challenge in conventional photocatalytic technology has been low energy conversion efficiency. Furthermore, in reactions under light irradiation, the light absorbed by the photocatalyst is converted into charge separation and heat (hot spots), which depend complexly on the intensity and temperature of the irradiated ultraviolet-visible light, making the reaction pathway for CO₂ reduction unclear.
1. To investigate the CO₂ reduction reaction pathway, the developed Ni–Ru–ZrO2 catalyst was compared with a conventional Ni–ZrO2 catalyst. Under conditions of light intensity of 568 milliwatts (mW/cm²) per cm² (Note 3), CO₂ photoreduction reaction tests were conducted with and without water cooling of the reactor (Fig. 1, 2).
2. At a light intensity of 654 mW/cm², the temperature of each Ni, Ru, and Zr atom was tracked under conditions with and without cooling by ethylene glycol (Note 4). Under cooling conditions, it was confirmed that light-generated electrons reduce CO₂ to COH, which is then accelerated 7-fold compared to the thermocatalytic reaction on the Ni surface at approximately 126°C, leading to reduction to methane (Fig. 2 left).
3. Under conditions without cooling, the Ni–Ru–ZrO2 catalyst produced methane at a high rate of over 9 millimoles per gram of catalyst per hour, with the reaction rate accelerated 2.7 times by the addition of Ru. Furthermore, density functional theory (DFT) calculations (Note 5) showed that Ru enabled CO₂ adsorption, allowing the methanation reaction to proceed solely through heating derived from light energy (Fig. 2 right).
■ Future Outlook
In this study, we improved the photocatalyst that converts CO₂ into fuel (methane) using light energy, scientifically demonstrating that the Ni–Ru–ZrO2 catalyst achieves the world's leading rate of CO₂ photo-methanation and proving the catalytic role of hot spots in metal nanoparticles. Moving forward, we aim to further enhance the efficiency of sustainable CO₂ utilization technologies using sunlight, such as for the synthesis of C2, C3 compounds (Note 6) and alcohols.
■ Glossary of Terms
Note 1) Light-generated electrons: Charge separation is called the separation of negative charges (i.e., electrons) and positive charges within a substance when light or other energy is applied, increasing their distance. Before they recombine, the negative charges cause reduction reactions, and the positive charges cause oxidation reactions in the photocatalyst.
Note 2) Hot spots: Limited areas that become high-temperature due to light being absorbed at an atomic level localized within a substance.
Note 3) Milliwatts (mW/cm²): A Watt (W) is the energy (Joules) per second. 1 Joule = 1 Newton × meter. Here, it represents the energy irradiated per square centimeter per second.
Note 4) Ethylene glycol: A liquid with the chemical formula HOC2H4OH, used in antifreeze. It was used in this experiment because it generates fewer bubbles than a water bath when tracking the temperature of each atom using synchrotron X-ray beams.
Note 5) Density Functional Theory (DFT) calculation: A theory in quantum mechanics that represents the wave equation for electrons as a function of electron density, used to investigate stable structures, energies, and electronic states for systems with many electrons.
Note 6) C2, C3 compounds: Compounds containing 2 or 3 carbon atoms in their molecules. Specifically, this refers to ethane, propane, ethylene, and propylene.
■ Publication Information
Title: Charge Separation and/or Hot Spots: Clarification of Efficient CO2 Reduction over Ru–Ni Nanoparticles Compared to Photocatalysis on Ru–Ni–ZrO2 Composites
Authors: Masahito Sasaki, Tomoki Oyumi, Keisuke Hara, Hongwei Zhang, and Yasuo Izumi
Journal: Journal of the American Chemical Society
DOI: 10.1021/jacs.5c17533
■ About the Research Project
This research was conducted with the following support:
・ Grant-in-Aid for Scientific Research (B) "Flexible and Precise Control of CO₂ to Various C2,3 Products Using Unsaturated Semiconductor-Metal Nanoparticle Photocatalysts" (24K01522)
・ Grant-in-Aid for Scientific Research (B) "Time-Resolved Tracking of CO₂ Photo-Multielectron Reduction and Isotope-Labeled Species on the Surface of Alloy Nanoparticles-Ultrathin Semiconductor Composites" (20H02834)
*Please check the PDF below for details.
d15177-1152-40864be57fd5c80d34bf4fe7485e7856.pdf