Berry to Exhibit 'QMSmart,' an eQMS Developed from a Field Perspective by a Medical Device Manufacturer, at Medtec Japan 2026
Berry will showcase 'QMSmart,' a cloud-based eQMS developed from a field perspective by a medical device manufacturer, at Medtec Japan 2026, Asia's largest exhibition for medical device manufacturing and design.
📋 Article Processing Timeline
- 📰 Published: April 2, 2026 at 20:00

Berry Inc. (Headquarters: Taito-ku, Tokyo; Representative Director: Hiroshi Nakano) will exhibit at "Medtec Japan 2026" (organized by Informa Markets Japan Co., Ltd.), to be held from April 21 (Tuesday) to 23 (Thursday), 2026, at Tokyo Big Sight East Hall 7. At the booth, we will conduct live demonstrations of 'QMSmart,' a cloud-based eQMS for medical device manufacturers.
Medtec Japan is a specialized exhibition for the medical device industry, where suppliers involved in medical device design and manufacturing gather with development and research personnel from domestic and international medical device manufacturers. This year marks the 17th edition, serving as a venue for product exhibitions, business negotiations, and information exchange, attracting many quality control, manufacturing, and R&D personnel active on the front lines of the medical device industry. This exhibition, which brings together companies of various sizes from ventures to mid-sized manufacturers, is a valuable opportunity for small and medium-sized medical device manufacturers facing QMS challenges to gather the latest information.
QMSmart is an eQMS developed by Berry itself to solve the challenges of "limitations of paper management" and "high cost of existing eQMS" that Berry faced in its own medical device manufacturing and sales operations. With an affordable pricing structure starting from 100,000 yen per month and ease of use utilizing AI, it supports startups and small and medium-sized medical device manufacturers in solving the challenges of QMS task individualization and personnel shortages.
■Highlights of the Berry Booth
You can experience the following at the booth:
・ Live demonstration of QMSmart: You can actually operate key functions such as document management, CAPA management, and training management to concretely confirm the operational image after introduction.
・ Live experience of AI functions: We will demonstrate AI-driven business automation, such as regulatory compliance checks for QMS ordinances and ISO13485, and automatic root cause analysis.
・ Introduction and pricing consultation: We will explain pricing plans starting from 100,000 yen per month and allow you to directly consult on the optimal introduction method tailored to your company's workflow.
■Main Features of QMSmart
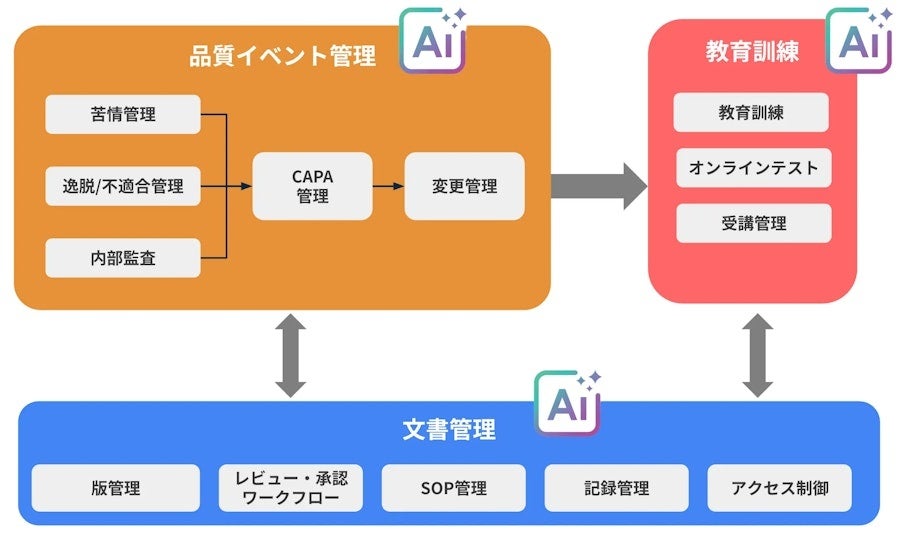
-
Document Management
・ All documents, from regulations and SOPs to complaint handling forms and other records, are digitized.
・ Instantly access necessary information with keyword search.
・ Compliant with electronic signatures, user permission management, and audit logs. -
Event Management
・ Centralized management of quality events such as CAPA on the cloud.
・ Progress status, related records, and history of various events are visible at a glance.
・ Flexible event templates support all quality events. -
Training Management
・ Centralized management from planning to attendance management of training linked to document revisions.
・ Significantly reduces the effort of training with automatic scoring of online tests and monitoring of attendance status.
・ Easily confirm attendance records for each participant. -
AI Support Functions
・ AI regulatory check for documents: Automatically checks regulatory compliance with QMS ordinances, ISO13485, etc.
・ Automatic root cause analysis: Deepens the analysis of root causes based on quality information such as complaints and past similar case data.
・ Automatic online test generation: Automatically generates online test questions from documents, significantly reducing the time for creating training materials.
■Exhibition Overview

|
Name |
Medtec Japan 2026 (17th Edition) |
|
Dates |
April 21 (Tuesday) - 23 (Thursday), 2026, 10:00 - 17:00 |
|
Organizer |
Informa Markets Japan Co., Ltd. (Medtec Japan Secretariat) |
|
Venue |
Tokyo Big Sight East Exhibition Hall 7 |
|
Booth Number |
605 |
|
URL |
About Berry Inc.
Berry Inc. is a medical device venture aiming to realize a society where "everyone can receive necessary medical care when needed." Utilizing 3D printing technology and 3D data analysis, we manufacture and sell "Baby Band," a cranial orthosis helmet for infants. Based on our experience in our own development and manufacturing sites, we are working to solve challenges in the medical device industry.
Furthermore, our company has acquired the international standard "ISO 27001" for information security management systems (ISMS), which recognizes our system for managing information confidentiality, integrity, and availability in a balanced manner. We are continuously working to build a safe and secure service provision system.
Established: July 2021
Location: Sumitomo Fudosan Ueno Okachimachi Building 4F, 3-7-1 Motomachi, Taito-ku, Tokyo
Representative Director: Hiroshi Nakano
Business Activities: Manufacturing and sales of medical devices / Design and development of software
Corporate Site: https://www.berryinc.co.jp/
Yakuji Navi: https://yakuji-navi.com/
FAQ
What kind of companies does QMSmart target?
QMSmart primarily targets medical device manufacturers from startups to mid-sized companies, especially SMEs struggling with QMS task individualization and personnel shortages.
What are the main features of QMSmart?
It includes document management, event management (e.g., CAPA), training management, and AI-powered support features like regulatory compliance checks, root cause analysis, and automatic online test generation.
What can be experienced at Medtec Japan 2026?
Visitors can experience live demonstrations of QMSmart, AI features, and consult on implementation and pricing. The booth number is 605.